In the fluorescent glare of the briefing room, the hum of phones buzzing in pockets and the low murmur of reporters packed in tight, the news was delivered with a gravity that silenced the room. "Over 3.1 million bottles of eye drops have been recalled nationwide," said the FDA spokesperson, their voice cutting through the tension. "This recall affects products sold at major retailers like Walgreens and CVS." The reason? A "lack of assurance of sterility," a phrase that sent a shiver through the room.
Understanding the FDA Eye Drop Recall
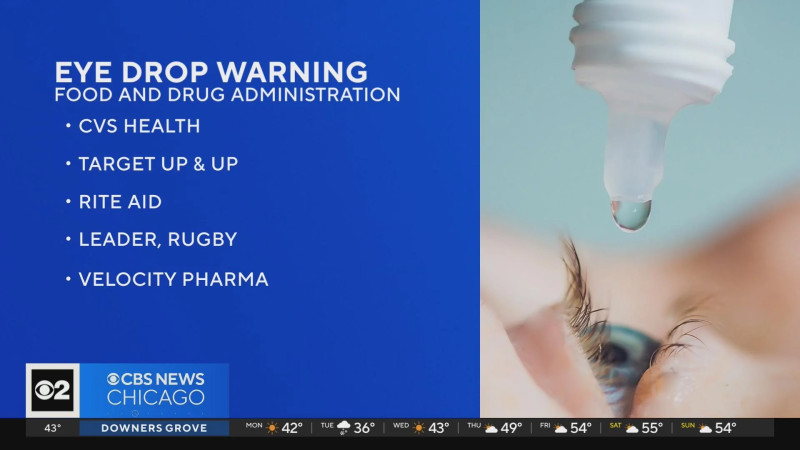
The Food and Drug Administration (FDA) has issued a voluntary recall affecting millions of eye drop products sold at major retail stores nation-wide, including Walgreens and CVS. The recall, which impacts over 3.1 million bottles, is due to concerns over sterility. The eye drops, manufactured by KC Pharmaceuticals Inc., were sold under various brand names and distributed by retailers such as Kroger, HEB, Meijer, and Military Exchanges. The recall comes in response to the potential risk of eye infections that could result in partial blindness or other severe complications.
The FDA's decision to issue a recall stems from the potential risk posed by non-sterile eye drops. According to the FDA, "the potential risk of eye infections that could result in partial blindness or other severe complications" is a significant concern. This recall underscores the importance of ensuring the sterility of over-the-counter eye drop products. The FDA's warning highlights the potential for severe health risks, including eye infections that could lead to partial blindness.
What Consumers Need to Know
Consumers are urged to check their medicine cabinets and avoid using any of the recalled eye drop products. The recall involves a wide range of brands distributed by major retailers, including CVS, Walgreens, Kroger, HEB, and Meijer, among others. The FDA has provided a comprehensive list of the affected products, which includes eye drops sold under various brand names.
To protect their health, consumers should take the following steps: - Do not purchase or use any of the recalled eye drop products. - Check the list of recalled products to ensure that any eye drops in their possession do not match the recalled items. - Return any recalled products to the place of purchase for a refund. - Consult a healthcare provider if they have used any of the recalled products and experience symptoms such as redness, swelling, or pain in the eye.
In a statement, an industry expert emphasized the importance of consumer vigilance: "Consumers need to be proactive in checking their products against the recall list. The risks associated with non-sterile eye drops are serious and can lead to severe health complications." — FDA Spokesperson
The Impact on Retailers and Manufacturers
The recall has significant implications for both retailers and manufacturers. Major retailers like Walgreens, CVS, Kroger, and HEB are now tasked with removing the recalled products from their shelves and providing refunds to customers. The recall also raises questions about the manufacturing processes and quality control measures at KC Pharmaceuticals Inc., the company behind the recalled products.
The recall highlights the broader issue of product safety in the pharmaceutical industry. As consumers become more aware of potential health risks, companies are under increasing pressure to ensure the sterility and safety of their products. The FDA's recall serves as a reminder of the importance of stringent quality control measures and the potential consequences of failing to meet these standards.
The FDA's advisory and subsequent recall of over 3.1 million eye drop bottles sold at Walgreens, CVS, and other major retailers marks a pivotal moment in consumer safety. As the public grapples with the implications of this massive recall, the focus remains on ensuring the sterility and safety of over-the-counter eye drop products, a key concern for consumers nationwide. The FDA’s vigilance and the industry's response will shape the future of product safety, with consumer trust hanging in the balance.