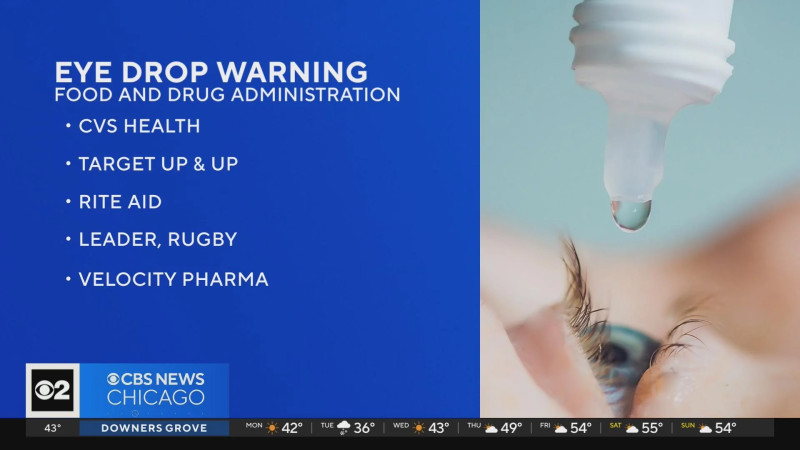
In a move that has sent ripples through the healthcare community, a eye drop recall in 2026 involving millions of bottles has been announced. The recall, initiated by the U.S. Food and Drug Administration (FDA), is due to concerns over sterility issues, raising significant questions about patient safety and regulatory oversight.
The Scale and Scope of the Eye Drop Recall 2026
The recall in 2026 is not just about a few isolated products; it spans multiple brands and a staggering number of bottles. More than 3 million bottles of eye drops have been pulled from the market, affecting major retailers like CVS, Walgreens, Kroger, and HEB. This widespread action underscores the severity of the issue, with regulators and consumers alike grappling with the implications. The recall includes eight different types of eye drop products, each distributed under various brand names. This means that millions of consumers who rely on these products for their eye health may be affected. Look, this isn't just about a few bottles here and there. This is a major public health concern that affects millions of Americans who depend on these eye drops for their daily eye care.
Understanding the Sterility Concerns
The core issue behind the massive eye drops recall is the potential lack of sterility in the products. Sterility is paramount in eye care products because any contamination can lead to severe infections and complications, including blindness. The FDA's enforcement report highlights that these products may not meet the necessary sterility standards, posing a significant risk to users. The FDA has classified this recall as a Class II, which means that using these products could cause temporary or medically reversible adverse health consequences.
Consumer Action and Safety Measures
So, what should you do if you or a loved one has been using these eye drops? Check your medicine cabinet immediately. If you find any of the recalled products, do not use them. The FDA and the affected companies have provided lists of the recalled products and their lot numbers. Visit the FDA's official website or the websites of the retailers involved to verify if your product is part of the recall. And then — get this — if you're unsure, play it safe and dispose of the product. Remember, your eye health is not something to gamble with.
If you've been using these eye drops and experience any symptoms such as redness, itching, or discharge, seek medical attention promptly. Early intervention can prevent more severe complications.“The FDA is committed to protecting public health through robust oversight of medical products. We urge consumers to check their eye drops and follow the recall guidelines to ensure their safety.” - FDA Statement
The Regulatory Response and Future Implications
This recall is a stark reminder of the rigorous standards that pharmaceutical companies must meet to ensure consumer safety. The FDA's swift action in recalling these products is a testament to its commitment to public health, but it also raises questions about the oversight and quality control measures in place for eye care products. What kind of regulatory lapses allowed this to happen? How can we prevent such widespread recalls in the future? As consumers, we trust that the products we use are safe and effective. This recall shakes that trust and demands a closer look at the systems in place to protect us.
In the meantime, the eyedrop recall serves as a wake-up call for both consumers and regulators. The FDA’s actions are a crucial step in addressing this issue, but the broader implications for the pharmaceutical industry and consumer trust are profound. Whether this incident will lead to stricter regulations and better quality control measures remains to be seen. But one thing is clear: the safety of consumer products, especially those affecting our health, should never be compromised. The next time you reach for an eye drop, you might want to think twice. Is there a safer alternative? And how can we ensure that these incidents don't happen again?