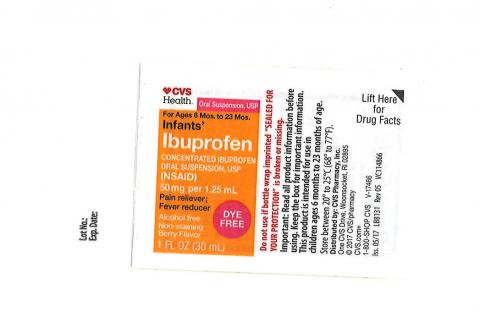
In the bustling streets of New York, the news is spreading like wildfire: nearly 90,000 bottles of children's oral ibuprofen have been recalled nationwide due to contamination concerns. The gel-like mass and black particles found in the products have prompted a swift response from the U.S. Food and Drug Administration (FDA) .
What Parents Need to Know About the Ibuprofen Recall
Parents are being urged to check their medicine cabinets immediately. The recall affects Children's Ibuprofen Oral Suspension, USP, a widely used medication for managing pain and inflammation in children. Stated one pharmacist at a local drug store, "We've had a lot of anxious parents coming in today, checking their labels and wanting to know more about the recall." The voluntary recall was initiated by Strides Pharma, Inc. after receiving multiple complaints from consumers .
Understanding the Risks and Precautions
An NSAID, or non-steroidal anti-inflammatory drug, ibuprofen is commonly used to reduce fever and relieve mild to moderate pain. Unlike Children's Tylenol, which works by increasing the body's tolerance for pain, ibuprofen targets inflammation specifically. The presence of foreign substances in the product raises significant safety concerns, especially for children who are particularly vulnerable to such contaminants .
In response to the recall, Strides Pharma, Inc. has issued a statement emphasizing the importance of consumer vigilance. The FDA is also urging parents and caregivers to check their medicine cabinets for any affected bottles and to report any adverse reactions or findings to the FDA's MedWatch program .
One mother, Lisa Johnson, shared her experience, "I had no idea until my pharmacist called me this morning. I'm glad I checked; it's terrifying to think what could have happened if I hadn't." The recall serves as a stark reminder of the ongoing need for vigilance in ensuring the safety of over-the-counter medications .
The Impact on Drug Safety and Regulation
The recall of nearly 90,000 bottles of ibuprofen is part of a broader pattern of quality and safety lapses in children's and over-the-counter drug products. Recent months have seen a series of similar recalls, prompting increased scrutiny from federal regulators .
The FDA has provided a searchable list of recalled products, allowing consumers to stay informed and take necessary precautions. As of now, there have been no reported injuries or adverse effects attributed to the contaminated ibuprofen, but the potential risk remains high .
Federal regulators are continuing to investigate the source of the contamination. Strides Pharma, Inc. has assured the public that it is cooperating fully with the FDA to address the issue and prevent future occurrences. "We take the safety of our products very seriously," a representative from Strides Pharma stated .
In the midst of the recall, many parents are questioning the overall safety of over-the-counter medications. "It's scary to think that something we trust to help our kids could be contaminated," said Jane Thompson, a local parent. The recall has sparked a renewed focus on drug safety and the importance of regulatory oversight .
As the community grapples with the implications of the recall, the atmosphere in local pharmacies and clinics is one of heightened caution and concern. Some parents have expressed their frustration, while others remain hopeful that the recall will lead to stricter safety measures in the future. As one parent put it, "We just want to know that the medications we’re giving our kids are safe."
"The ibuprofen recall highlights the ongoing challenges in ensuring the safety of over-the-counter medications. The way regulatory concerns can instill anxiety in communities echoes past crises, such as the Tylenol scare in the 1980s .