Parents stand on alert: nearly 90,000 bottles of children’s ibuprofen, a staple in many medicine cabinets, have been recalled. The U.S. Food and Drug Administration (FDA) has issued a nationwide recall of Children's Ibuprofen Oral Suspension, USP, following reports of contamination and foreign substances. Think again before you reach for that trusted bottle.
FDA Issues Nationwide Recall of Children's Ibuprofen
The FDA announced the voluntary recall on March 2, 2026, after Strides Pharma, the manufacturer, received numerous complaints about a gel-like mass and black particles in the medication.
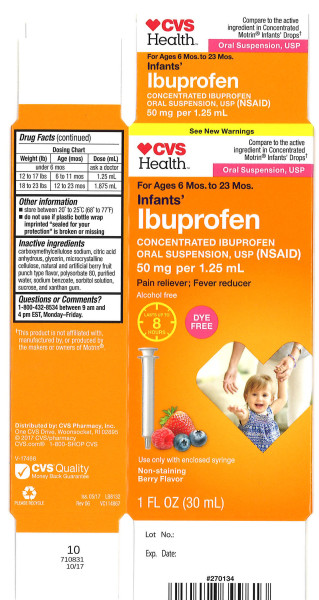
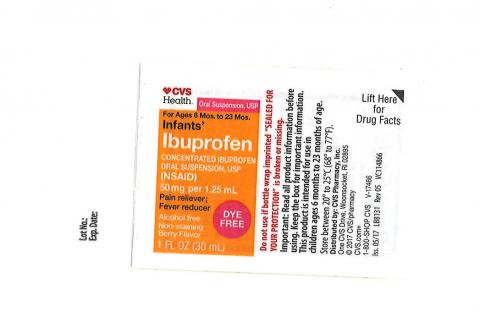
This ibuprofen recall affects 89,592 bottles of the Children's Ibuprofen Oral Suspension, USP, 100 mg per 5 mL, sold in 4-ounce (120 mL) bottles under the NDC 51672-5321-8. Gone. That familiar bottle, now a potential health risk.
Strides Pharma initiated the recall to ensure safety, urging consumers to check their medicine cabinets and avoid using the affected products.
“This recall underscores the importance of vigilance in pharmaceutical manufacturing and distribution to protect public health.[Strides Pharma] is committed to ensuring the safety and quality of our products, and we are taking immediate corrective actions to address this issue,” Strides Pharma said in a statement.
Parents Urged to Check Medicine Cabinets for Contaminated Ibuprofen
Parents are advised to check their medicine cabinets for the affected bottles. The recall includes specific lot numbers and expiration dates, which have been listed by the FDA as part of its public safety announcement. This is where you come in. Double-check that bottle before giving it to your child. This recall does not impact other Ibuprofen products or different formulations, such as tablets or capsules.
“This is a critical reminder for parents and caregivers to always inspect the medications they administer to their children.”. “If you have any questions or concerns, or if you suspect you’ve been affected, contact your healthcare provider or the FDA immediately.”
What to Do if You Have the Recalled Ibuprofen
If you have the recalled product, do not use it. Take it back to the store where you purchased it for a refund or dispose of it according to local regulations for medication disposal. Strides Pharma and the FDA are working together to manage this recall, ensuring all affected products are removed from circulation.
Consumers should contact Strides Pharmaceuticals at 1-877-877. The FDA is notifying retailers to remove the product from their shelves. This may affect other brands as well, as the product is distributed nationally.
“The FDA is committed to protecting public health and ensuring the safety of medications.”[FDA] is monitoring the situation closely and taking all necessary actions to address the issue.
”The FDA is committed to protecting public health and ensuring the safety of medications.”[FDA] is monitoring the situation closely and taking all necessary actions to address the issue.
The FDA’s announcement of the children’s ibuprofen recall 2026 serves as a reminder of the importance of vigilance in monitoring the safety of medications, especially those used by our most vulnerable populations.
We have a staggering number of affected households, a potential crisis in countless homes, a silent illness waiting to strike. This is where the children’s ibuprofen recall 2026 story begins. And it’s not over yet.